 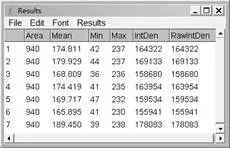
KDM5A showed a similar higher level of mRNA expression in sarcomas (SARC) (Fig. KDM5A was upregulated in malignant osteosarcoma tissues Finally, this study provided a deeper understanding of the molecular mechanism of KDM5A underlying the osteosarcoma progression. Moreover, the potential role of KDM5A on the proliferation of osteosarcoma was investigated. In the present work, we examined the expression of KDM5A among human osteosarcoma tissues and its relationship with the clinical characteristic patients. However, the expression of KDM5A and its clinical significance in osteosarcoma remains unclear. KDM5A was also considered as a potential therapeutic target in small-cell lung cancer 12, glioblastoma (GBM) 13, 14, hepatocellular carcinoma 15, breast cancer 16, and renal cell carcinoma 17. KDM5A has been reported to be highly expressed in ovarian cancer tissues and especially in SKOV3/paclitaxel (PTX) cells, which were resistant to PTX 11. Misregulation of KDM5A contributed to the pathogenesis of lung and gastric cancers 10. Mutations in tumor suppressor genes, as RB1 7, 8 and p53 gene ( TP53), were strongly associated with human osteosarcoma 9. KDM5A was originally identified as a transcriptional repressor of pRB (protein of retinoblastoma, RB1) and played a role in differentiation of neural progenitor cells 4, 5, 6. Therefore, exploring the novel molecular mechanism of tumor growth could help develop a new therapeutic intervention to improve overall outcome in patients with malignant osteosarcoma 3. Uncontrollable cell proliferation was one of the main factors in the carcinogenesis and development of osteosarcoma 3. Unfortunately, although modern treatment protocols combined with chemotherapy, surgery, and radiotherapy, the metastasis and recurrence of the osteosarcoma contributed mainly to the poor prognosis of patients 2. Osteosarcoma was one of the most common primary malignancies arising from bone and affects primarily children, adolescents, and young adults 1. Taken together, our research established a role of KDM5A in osteosarcoma tumorigenesis and progression. RNA-Seq of KDM5A-KO cells indicated that interferon, epithelial–mesenchymal transition (EMT), IL6/JAK/STAT3, and TNF-α/NF-κB pathway were likely involved in the regulation of osteosarcoma cell viability. Furthermore, after knockout of KDM5A in osteosarcoma cells by CRISPR/Cas9 system, the tumor size and growth speed were inhibited in tumor-bearing nude mice. Moreover, knockdown of KDM5A could increase the expression level of P27 (cell-cycle inhibitor) and decrease the expression of Cyclin D1. Knockdown of KDM5A suppressed osteosarcoma cell proliferation and induced apoptosis. In the current study, KDM5A was highly expressed in osteosarcoma than adjacent normal tissue. However, the function of KDM5A in the carcinogenesis of osteosarcoma is not clear. Previous studies showed that histone demethylase KDM5A can increase the proliferation and metastasis of several cancers. 
Therefore, it is of vital importance to explore novel prognostic molecular biomarkers and targets for more effective therapeutic approaches. Osteosarcoma is a primary bone malignancy with a high rate of recurrence and poorer prognosis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
